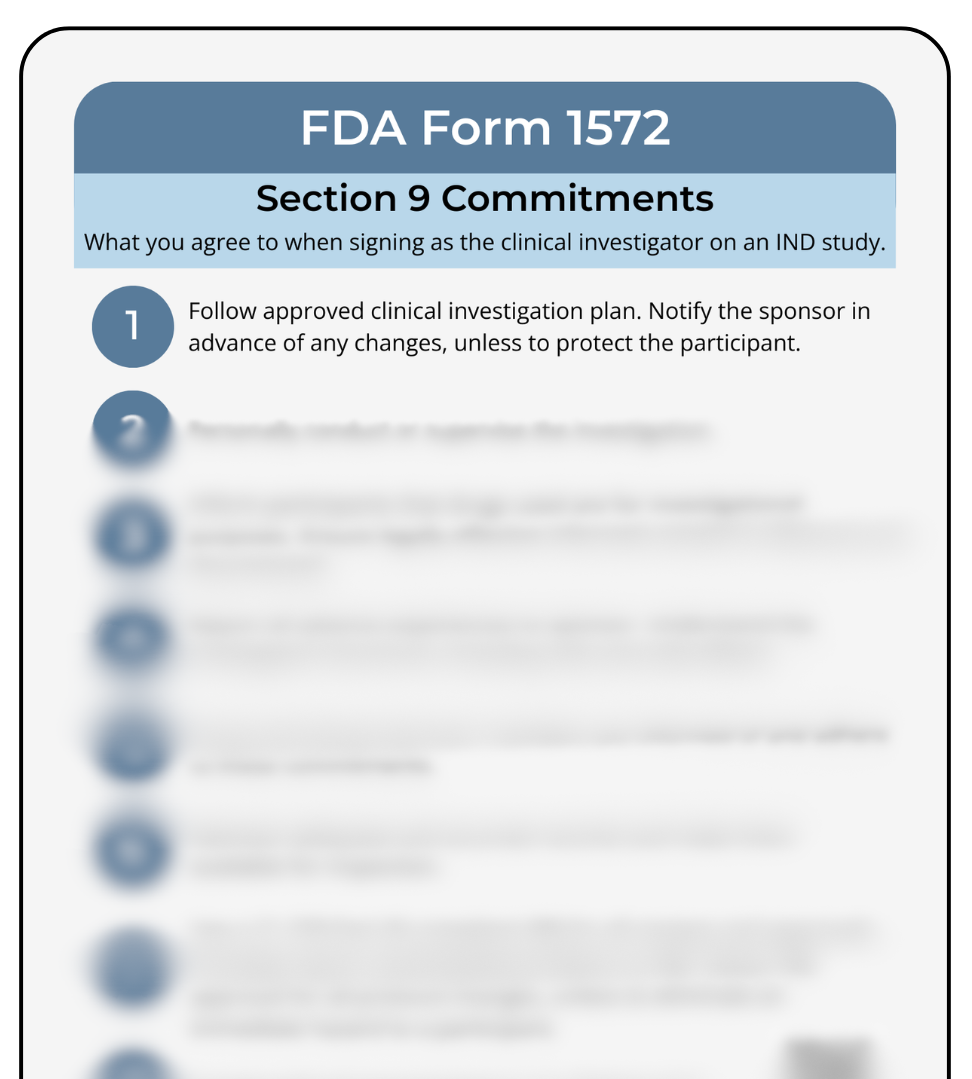
Break down what the FDA requires when investigators sign Form 1572 — including adherence to the investigational plan, informed consent, accurate documentation, safety reporting, and more.
Use this reference to reinforce expectations around supervision, informed consent, safety reporting, IRB oversight, and recordkeeping — all grounded in 21 CFR 312 requirements.
A practical tool for sponsors, investigators, and site staff to ensure everyone understands the same commitments the same way — consistent, transparent, and easy to reference anytime.
Have every Form 1572 commitment in one clear, accessible place.
When investigators sign the FDA Form 1572, they commit to a defined set of responsibilities that guide the conduct of the clinical investigation. This one-page reference pulls every Section 9 commitment into a clean, easy-to-review format so investigators, sponsors, coordinators, and students can quickly revisit what the FDA requires under Form 1572 and 21 CFR 312. Whether you’re onboarding a new investigator, supporting a study team, training students, or preparing for monitoring visits, this sheet helps everyone stay aligned on expectations around supervision, informed consent, IRB oversight, recordkeeping, protocol compliance, and safety reporting. It’s a practical addition to investigator packets, regulatory binders, sponsor training materials, and site onboarding workflows. Licensed for individual or single-site use. Full licensing details can be found at catalog.cliniq.academy/pages/license-agreement.
Keep responsibilities clear and accessible.
A straightforward reminder of the commitments tied to FDA Form 1572 — easy to reference, easy to share, and ready for real-world study work.
$5.00